About the Test
Introduction
The Coronavirus disease (COVID-19) is an infectious disease caused by a newly discovered coronavirus, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2)1. The SARS-CoV-2 is a β-coronavirus, which is an enveloped non-segmented positive-sense RNA virus2. It is spread by human-to-human transmission via droplets or direct contact, and infection has been estimated to have a mean incubation period of 6.4 days and a basic reproduction number of 2.2 4-3.58. Among patients with pneumonia caused by SARS-CoV-2, fever was the most common symptom, followed by cough3 The main IVD assays used for COVID-19 employ real-time reverse transcriptase-polymerase chain reaction (RT-PCR) that takes a few hours4 . The availability of a cost-effective, rapid pointof-care diagnostic test is critical to enable healthcare professionals to aid in the diagnosis of patients and prevent further spread of the virus5 . Antigen tests will play a critical role in the fight against COVID-196
.
Test Principle
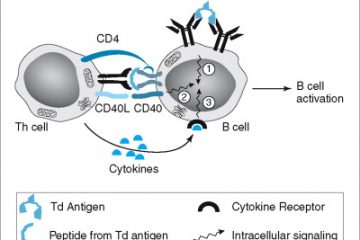
Panbio™ COVID-19 Ag Rapid Test Device contains a membrane strip, which is pre-coated with immobilized anti-SARS-CoV-2 antibody on the test line and mouse monoclonal anti-chicken IgY on the control line. Two types of conjugates (human IgG specific to SARS-CoV-2 Ag gold conjugate (binds to the nucleocapsid
protein) and chicken IgY gold conjugate) move upward on the membrane chromatographically and react with anti-SARS-CoV-2 antibody and pre-coated mouse monoclonal anti-chicken IgY respectively. For a positive result, human IgG specific to SARS-CoV-2 Ag gold conjugate and anti-SARS-CoV-2 antibody will form a test line in the result window. Neither the test line nor the control line are visible in the result window prior to applying the patient specimen. A visible control line is required to indicate a test result is valid. Intended Use Panbio COVID-19 Ag Rapid Test Device is an in vitro diagnostic rapid test for the qualitative detection of SARS-CoV-2 antigen (Ag) in human nasal swabspecimens from individuals who meet COVID-19 clinical and / or epidemiological criteria.
Panbio™ COVID-19 Ag Rapid Test Device is for professional use only and is intended to be used as an aid in the diagnosis of SARS-CoV-2 infection. The product may be used in any laboratory and non-laboratory environment that
meets the requirements specified in the Instructions for Use and local regulation. The test provides preliminary test results. Negative results don’t preclude SARSCoV-2 infection and they cannot be used as the sole basis for treatment or other management decisions. Negative results must be combined with clinical observations, patient history, and epidemiological information. The test is not intended to be used as a donor screening test for SARS-CoV-2.
Antibody tests to expand testing and help healthcare workers
While molecular testing detects whether someone has the virus, antibody tests determine if someone was previously infected.
Abbott’s SARS-CoV-2 IgG test identifies the IgG antibody, which is a protein that the body produces in the late stages of infection and may remain for up to months and possibly years after a person has recovered. Abbott’s IgG antibody test will initially be available on its ARCHITECT i1000SR and i2000SR laboratory instruments*. More than 2,000 of these instruments are in use in U.S. laboratories. These instruments can run up to 100-200 tests per hour.
Abbott is making the test available as part of the U.S. Food and Drug Administration (FDA) notification without an Emergency Use Authorization (EUA) pathway outlined in Policy for Diagnostic Tests for Coronavirus Disease-2019 during the Public Health Emergency. Additionally, Abbott plans to file an EUA submission with the FDA and plans to CE Mark to the IVD Directive (98/79/EC) in the European Union.
Abbott is significantly scaling up its manufacturing for antibody testing and is expecting to immediately ship close to 1 million tests this week to U.S. customers, and will ship a total of 4 million tests in total for April. The company is ramping up to 20 million tests in the U.S. in June and beyond as it expands the tests to run on its new Alinity™ i system. Abbott also will be expanding its laboratory antibody testing to the detection of the antibody, IgM, in the near future.
[Linking template=”default” type=”products” search=”lgm rapid test device” header=”3″ limit=”28″ start=”1″ showCatalogNumber=”true” showSize=”true” showSupplier=”true” showPrice=”true” showDescription=”true” showAdditionalInformation=”true” showImage=”true” showSchemaMarkup=”true” imageWidth=”” imageHeight=””]