Interleukin(IL)-Four is produced by T cells and different leukocytes and is a crucial mediator of monocyte and B cell responses. During routine circulation cytometry panel validation for the investigation of intracellular cytokines, we noticed distinctive IL-4 expression patterns related to the broadly obtainable monoclonal antibody 8D4-8.
Namely, IL-4 (8D4-8) expression was noticed within the absence of mobile activation and enhanced following staurosporine publicity. Mass spectrometry evaluation of immunoprecipitates from peripheral blood lymphocytes (PBL) revealed that 8D4-8 cross-reacts with the ever-present cytoskeletal protein myosin-9.
We confirmed these outcomes by western blotting immunoprecipitates, utilizing immunofluorescence amongst staurosporine-treated Caco-2 cells, and by surface-labeling PBL for 8D4-8 and myosin-9 and analyzing by circulation cytometry. Although beforehand reported from a number of impartial teams, we discovered no proof to help the speculation that IL-4 is produced by apoptotic cells. Rather, this seems to have been myosin-9. Our information point out clone 8D4-8 shouldn’t be used within the circulation cytometric examine of IL-4. Furthermore, our work calls for a reevaluation of earlier circulation cytometric research which have used this clone for IL-4 evaluation and highlights the significance of validation in antibody-based assays.
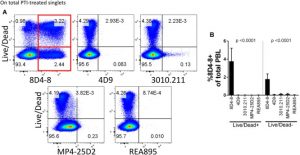
Anti-human Interleukin(IL)-4 Clone 8D4-8 Cross-Reacts With Myosin-9 Associated With Apoptotic Cells and Should Not Be Used for Flow Cytometry Applications Querying IL-4 Expression.
Anti-EGFR-resistant clones decay exponentially after development: implications for anti-EGFR re-challenge.
<AbstractText>Colorectal most cancers (CRC) has been proven to accumulate RAS and EGFR ectodomain mutations as mechanisms of resistance to epidermal progress issue receptor (EGFR) inhibition (anti-EGFR). After anti-EGFR withdrawal, RAS and EGFR mutant clones lack a progress benefit relative to different clones and decay; nonetheless, the kinetics of decay stay unclear.
We sought to find out the kinetics of acquired RAS/EGFR mutations after discontinuation of anti-EGFR remedy.</AbstractText><AbstractText>We current the post-progression circulating tumor DNA (ctDNA) profiles of 135 sufferers with RAS/BRAF wild-type metastatic CRC handled with anti-EGFR who acquired RAS and/or EGFR mutations throughout remedy. Our validation cohort consisted of an exterior dataset of 73 sufferers with a ctDNA profile suggestive of prior anti-EGFR publicity and serial sampling. A separate retrospective cohort of 80 sufferers was used to guage total response fee and development free survival throughout re-challenge therapies.</AbstractText><p><div><b>RESULTS</b></div>Our evaluation confirmed that RAS and EGFR relative mutant allele frequency decays exponentially (r<sup>2</sup>=0.93 for RAS; r<sup>2</sup>=0.94 for EGFR) with a cumulative half-life of 4.Four months.
We validated our findings utilizing an exterior dataset of 73 sufferers with a ctDNA profile suggestive of prior anti-EGFR publicity and serial sampling, confirming exponential decay with an estimated half-life of 4.three months. A separate retrospective cohort of 80 sufferers confirmed that sufferers had the next total response fee throughout re-challenge therapies after growing time intervals, as predicted by our mannequin.</p><AbstractText>These outcomes present scientific help for anti-EGFR re-challenge and information the optimum timing of re-challenge initiation.</AbstractText>
Primary and secondary anti-viral response captured by the dynamics and phenotype of particular person T cell clones.
The various repertoire of T-cell receptors (TCR) performs a key position within the adaptive immune response to infections. Using TCR alpha and beta repertoire sequencing for T-cell subsets, in addition to single-cell RNAseq and TCRseq, we observe the concentrations and phenotypes of particular person T-cell clones in response to major and secondary yellow fever immunization – the mannequin for acute an infection in people – exhibiting their massive variety. We affirm the secondary response is an order of magnitude weaker, albeit ∼ 10 days quicker than the first one.
[Linking template=”default” type=”products” search=”myd88″ header=”2″ limit=”160″ start=”2″ showCatalogNumber=”true” showSize=”true” showSupplier=”true” showPrice=”true” showDescription=”true” showAdditionalInformation=”true” showImage=”true” showSchemaMarkup=”true” imageWidth=”” imageHeight=””]
Estimating the fraction of the T-cell response directed in opposition to the one immunodominant epitope, we establish the sequence options of TCRs that outline the excessive precursor frequency of the 2 main TCR motifs particular for this explicit epitope. We additionally present the consistency of clonal growth dynamics between bulk alpha and beta repertoires, utilizing a brand new methodology to reconstruct alpha-beta pairings from clonal trajectories.